The universe is full of some pretty cool stuff. But what makes up that stuff? Our detailed periodic table of elements timeline maps the history of humanity’s incredible discoveries of the stuff that makes up stuff!
Here is a printable PDF of A Timeline of When Elements Were Discovered, feel free to share and print
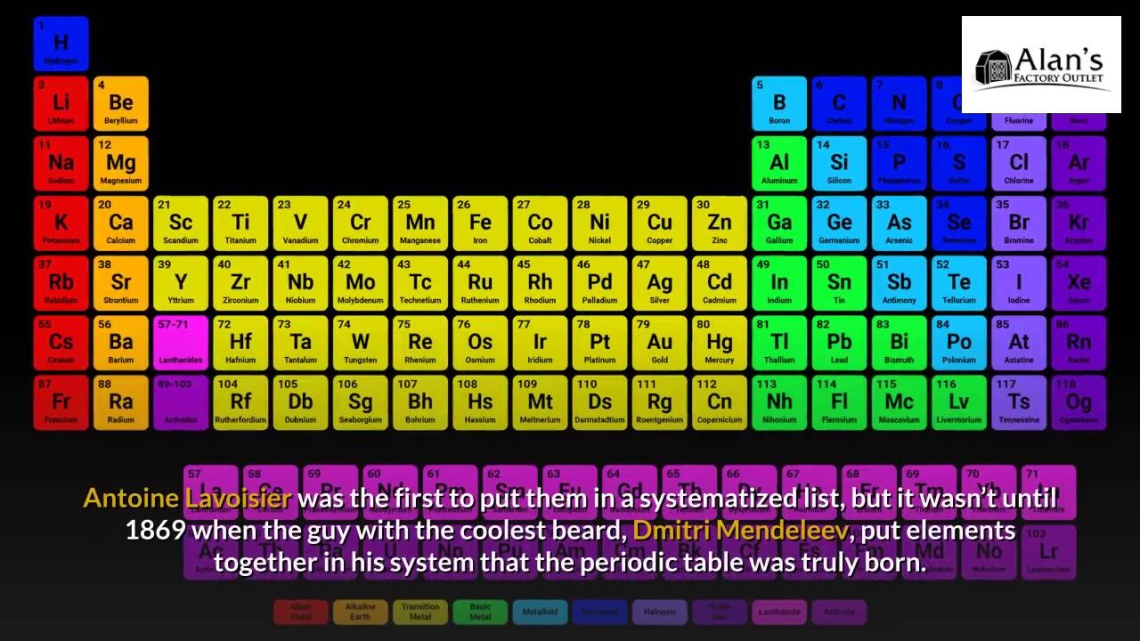
From the readily available copper to the noble gases hiding in the air we breathe, elements have been hiding in plain sight for eons. Chemical elements are the indivisible substances that make up the building blocks of matter — or, really, “stuff.” Made up of atoms with a certain number of protons, neutrons, and electrons, each one fits perfectly within its place on the periodic table of elements.
Timeline dates show us, though, that the journey of figuring all of this out has been a bumpy one. For one thing, our modern table wasn’t systematized until 1869 by Dmitri Mendeleev, whose table even featured blank spots for elements that hadn’t been discovered yet!
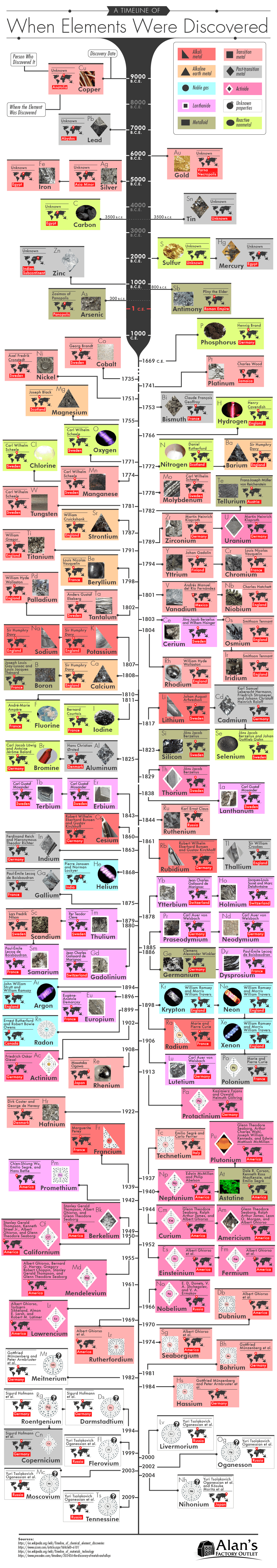
Element discovery dates range from ancient times to the modern era. Due to the availability of many common metal elements, our history of working with and using elements like gold, copper, and lead far predates the history of the periodic table. The timeline of the history of periodic table is sporadic, with some “discoveries” happening via ancient artifacts and others happening during a wave of chemistry discoveries in the 17th century. We also saw a huge explosion of scientific discovery of elements, for better and for worse, during the mid-20th century while bigger and bigger atoms were being worked with as part of nuclear weapons research. Check out the history of chemical elements and when they were discovered to see how our journey to understanding these combinations of protons, neutrons, and electrons has changed over the centuries!
The Discoverers of Periodic Elements
Click on the image to view full size

Here is a printable PDF of Discoverers of Periodic Elements, feel free to share and print
What Was the First Element Discovered?
While it was not understood to be an element at that time, the first element discovered was copper (Cu) due to the fact that its oldest known use was in 9,000 B.C.E. and the oldest existing sample from that era was from 6,000 B.C.E. Also in antiquity, lead, gold, silver, iron, tin, carbon, and several other elements commonly found in nature were “discovered.” This was long before Democritus and his teacher Leucippus conceptualized atomic theory among the ancient Greeks and long, long before anyone had attempted to arrange a periodic table of elements. Discovery dates after antiquity include those elements that were chemically found. The first of those chemically found discoveries was in 1669, when Henning Brand discovered phosphorus by boiling urine in his quest to discover the philosopher’s stone.
What Is the Most Recent Element Discovered?
The latest element discovered wasn’t so much “discovered” as it was synthesized: tennessine (Ts). A Russian-American collaboration created the element in 2009, officially announcing it in 2010. This most recently discovered element was one of several that have been synthesized in labs in the 21st century, with others including Nihonium (named after Japan), Moscovium (named after Moscow), Oganesson (named after scientist Yuri Oganessian), and Livermorium (named after a U.S. federal research laboratory).
Are We Still Discovering New Elements?
Many people believe the discovery of chemical elements has slowed down since the Manhattan Project in the 1940s, but this is not the case. No one knows how heavy we can go, but theoretically, elements 119 and 120 are possible with current technology. They are likely not found in nature and exceedingly difficult to create. They would also require a new row to be added to the periodic table.
When Were All the Elements Discovered?
Our infographic shows when each element was discovered, but here is our list of elements in order of discovery written out as well.
List of Elements by Discovery Date
| Copper Cu | 9000 B.C.E. |
| Lead Pb | 7000 B.C.E. |
| Gold Au | 6000 B.C.E. |
| Silver Ag | 5000 B.C.E. |
| Iron Fe | 5000 B.C.E. |
| Carbon C | 3500 B.C.E. |
| Tin Sn | 3500 B.C.E. |
| Sulfur S | 2000 B.C.E. |
| Mercury Hg | 2000 B.C.E. |
| Zinc Zn | 1000 B.C.E. |
| Arsenic As | 300 B.C.E. |
| Antimony Sb | 800 B.C.E. |
| Phosphorus P | 1669 C.E. |
| Cobalt Co | 1735 |
| Platinum Pt | 1741 |
| Nickel Ni | 1751 |
| Bismuth Bi | 1753 |
| Magnesium Mg | 1755 |
| Hydrogen H | 1766 |
| Oxygen O | 1771 |
| Nitrogen N | 1772 |
| Barium Ba | 1772 |
| Chlorine Cl | 1774 |
| Manganese Mn | 1774 |
| Molybdenum Mo | 1778 |
| Tungsten W | 1781 |
| Tellurium Te | 1782 |
| Strontium Sr | 1787 |
| Zirconium Zr | 1789 |
| Uranium U | 1789 |
| Titanium Ti | 1791 |
| Yttrium Y | 1794 |
| Chromium Cr | 1794 |
| Beryllium Be | 1798 |
| Vanadium V | 1801 |
| Niobium Nb | 1801 |
| Tantalum Ta | 1802 |
| Palladium Pd | 1802 |
| Cerium Ce | 1803 |
| Osmium Os | 1803 |
| Iridium Ir | 1803 |
| Rhodium Rh | 1804 |
| Potassium K | 1807 |
| Sodium Na | 1807 |
| Calcium Ca | 1808 |
| Boron B | 1808 |
| Fluorine F | 1810 |
| Iodine I | 1811 |
| Lithium Li | 1817 |
| Cadmium Cd | 1817 |
| Selenium Se | 1817 |
| Silicon Si | 1823 |
| Aluminum Al | 1825 |
| Bromine Br | 1825 |
| Thorium Th | 1829 |
| Lanthanum La | 1838 |
| Erbium Er | 1843 |
| Terbium Tb | 1843 |
| Ruthenium Ru | 1844 |
| Cesium CS | 1860 |
| Rubidium Rb | 1861 |
| Thallium Tl | 1861 |
| Indium In | 1863 |
| Helium He | 1868 |
| Gallium Ga | 1875 |
| Ytterbium Yb | 1878 |
| Holmium Ho | 1878 |
| Thulium Tm | 1879 |
| Scandium Sc | 1879 |
| Samarium Sm | 1879 |
| Gadolinium Gd | 1880 |
| Praseodymium Pr | 1885 |
| Neodymium Nd | 1885 |
| Germanium Ge | 1886 |
| Dysprosium Dy | 1886 |
| Argon Ar | 1894 |
| Europium Eu | 1896 |
| Krypton Kr | 1898 |
| Neon Ne | 1898 |
| Xenon Xe | 1898 |
| Polonium Po | 1898 |
| Radium Ra | 1898 |
| Radon Rn | 1899 |
| Actinium Ac | 1902 |
| Lutetium Lu | 1906 |
| Rhenium Re | 1908 |
| Protactinium Pa | 1913 |
| Hafnium Hf | 1922 |
| Technetium Tc | 1937 |
| Francium Fr | 1939 |
| Neptunium Np | 1940 |
| Astatine At | 1940 |
| Plutonium Pu | 1940 |
| Promethium Pm | 1942 |
| Curium Cm | 1944 |
| Americium Am | 1944 |
| Berkelium Bk | 1949 |
| Californium Cf | 1950 |
| Einsteinium Es | 1952 |
| Fermium Fm | 1952 |
| Mendelevium Md | 1955 |
| Lawrencium Lr | 1961 |
| Nobelium No | 1966 |
| Rutherfordium Rf | 1969 |
| Dubnium Db | 1970 |
| Seaborgium Sg | 1974 |
| Bohrium Bh | 1981 |
| Meitnerium Mt | 1982 |
| Hassium Hs | 1984 |
| Darmstadtium Ds | 1994 |
| Roentgenium Rg | 1994 |
| Copernicium Cn | 1994 |
| Flerovium Fl | 1999 |
| Livermorium Lv | 2000 |
| Oganesson Og | 2002 |
| Moscovium Mc | 2003 |
| Nihonium Nh | 2004 |
| Tennessine Ts | 2009 |
Our periodic table discovery timeline maps the journey of when we discovered some of the coolest metal substances ever. Most materials on the periodic table are some form of metal, and the discoveries of the periodic elements allowed us to create more and more powerful metals and alloys over time, enabling us to create stronger and harder metals. Who knows? Perhaps the volatile, atomically heavier elements we synthesize will let us build cooler and more amazing stuff!
This educational resource was put together by Alan’s Factory Outlet, makers of high-quality steel buildings and carports for sale.
Photo sources:
Photos by Jurii (Wikimedia.org)
Photos by Hi-Res Images of Chemical Elements (Wikimedia.org)
Photos by Robert M. Lavinsky (Wikimedia.org)
Photos by W. Oelen (Wikimedia.org)
Photos by Unknown (Wikimedia.org)
Original works by Greg Robson (Wikimedia.org)
“Gold (Nevada, USA) 1” photo by James St. John (Flickr)
“Element mercury (HG), liquid form” photo by Bionerd (Wikimedia.org)
Zosimos photo by Rvalette (Wikimedia.org)
“Crystals of pure platinum…” photo by Periodictableru (Wikimedia.org)
“Das chemische Element und Metall Wismut…” photo by Alchemist-hp (Wikimedia.org)
“A chunk of vapor-deposited magnesium crystals…” by Warut Roonguthai (Wikimedia.org)
Mangnanese photo by Thamizhpparithi Maari (Wikimedia.org)
“Hydrogen lamp” photo by UCL Mathematical & Physical Sciences (Flickr)
“Liquid nitrogren…” photo by Cory Doctorow (Flickr)
“I mixed bleach and muriatic acid…” photo by Larenmclane (Wikimedia.org)
“Niobium crystal…” photo by Dnn87 (Wikimedia.org)
“Cluster of osmium…” photo by Periodictableru (Wikimedia.org)
“Potassium pearls…” photo by Unknown (Wikimedia.org)
“Boron…” photo by Xvazquez (Wikimedia.org)
“crystals of flourite…” photo by Parent Gèry (Wikimedia.org)
“600px-Lanthanum-2” photo by GrrlScientist (Flickr)
“Erbium…” photo by Tomihahndorf (Wikimedia.org)
“Cesium/Caesium…” photo by Dnn87 (Wikimedia.org)
“Gallium crystals..” photo by en:user:foobar (Wikimedia.org)
“Crystal of Scandium…” photo by JanDerChemiker (Wikimedia.org)
“enhanced Bohr model…” original work by Ahazard.sciencewriter (Wikimedia.org)
“Astatine…” photo by Elahe81 (Wikimedia.org)
“A small disc of Am-241…” photo by Bionerd (Wikimedia.org)